 
It is a straightforward way of looking at the second law of thermodynamics. Many seek to view this law from the lens of the Clausius statement.īut what is the Clausius statement? The Clausius Statement 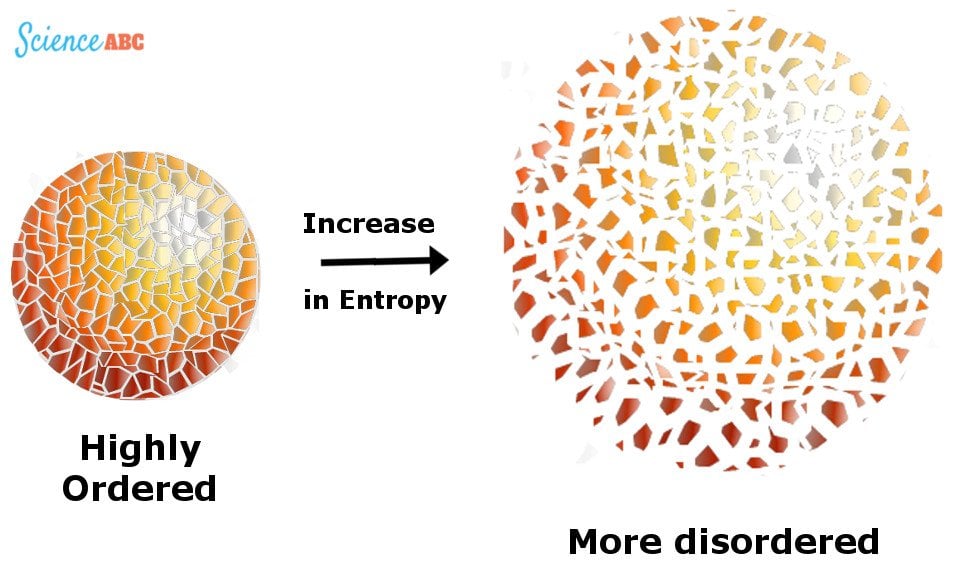 
One can view this aspect in several fundamental ways. This means that it has no variations in points across each aspect that ranges from energy, matter, and even temperature. And frequently, a high probability of natural methods to move to a stage of homogeneity or a system where it has the same properties at each point. A core concept in thermodynamics is that natural processes are irreversible. Indeed, the second law of thermodynamics is quite fascinating it revolves around the idea of irreversibility. Remember that actions do not happen within a reversible process due to the arrow of time. As more activity and actions occur, more friction happens, heat generates, and we end up with more irreversible process points. Due to more interactions, the second law of thermodynamics states that there will be more entropy.Īs you can see from merely sitting or standing there and reading these words, you are releasing heat into the universe, adding interactions, and increasing entropy. It seems that the entropy of the universe is only increasing due to its many potential states. The idea of the irreversible process is a critical one for entropy change in the second law. That is to say that the molecules, once moving to different states, will continue to move around as opposed to clustering in a specific area or staying in one shape. If molecules continue to move around within an isolated system, that shows a high level of entropy.įurther, in many cases, due to these processes increasing within an isolated system, we will note that most closed systems have irreversible processes. Still, in many closed systems, various molecules move to different points within that system, increasing the level of entropy. For instance, if you had a closed system where molecules had to stay in a specific area and would not transfer elsewhere, you would state it has low entropy. It makes sense on the face of it because of movement. In the entropy of a system, we are talking about the disorder present within a system and, in essence, the various states that the isolated system could have. The entropy of the universe or closed thermodynamic system only increases one of the more popular statements in the second law of thermodynamics. The s econd law of thermodynamics states that “in all energy exchanges if no energy enters or leaves the system, the potential energy of the state will always be less than that of the initial state.” Here is what you need to know about the second law of thermodynamics and why it is even more relevant as we seek to solve the pressing challenges that we face today in New York, Los Angeles, and other cities and rural towns worldwide. From thermal equilibrium to a thermodynamic system and what it means for you, you must understand and refresh yourself on the law of thermodynamic states to understand temperature, energy, and building blocks of the universe. The first law of thermodynamics or the second law, or even other laws of thermodynamics, may not have seemed relevant at the time on a larger scale, but they relate to every aspect of life. It is important to consider if we are to move forward with energy conservation, sustainability processes and systems, and overall equilibrium. Individuals like Sadi Carnot, Rudolf Clausius, their German physicist colleagues, and other science members discussed maximum entropy, temperature, heat q, heat flows per unit, and other thermodynamic concepts. But it is an essential factor in the discussion of sustainability, efficiency, and measurement of progress. Yes, many people learn this at their university and understand how to measure gas or other aspects to conduct calculations within their university surroundings to pass their course and move on to the next part of their journey. At the same time, the mention of thermodynamics can make many people automatically mentally check out. There many laws that govern humanity, but none are intriguing as the universal laws, such as the law of entropy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
